THERAPEUTIC INDICATIONSEpipen Jr. auto injectors are automatic injection devices that contain adrenaline for allergic emergencies. The automatic injector is designed for children with a body weight of 7.5-25 kg. Automatic injectors should only be used by a person with a history or recognized risk of an anaphylactic reaction. Automatic injectors are indicated in the emergency treatment of allergic reactions (anaphylaxis) to insect bites or stings, food, drugs and other allergens, as well as idiopathic or exercise induced anaphylaxis. These reactions can occur minutes after exposure and consist of redness, apprehension, syncope, tachycardia, chronic or unreachable pulse associated with a drop in blood pressure, seizures, vomiting, diarrhea and abdominal cramps, involuntary urination, wheezing, spastic dyspnea Laryngeal, itching, rash, urticaria or angioedema.
0 Comments
INDICATIONSEpinephrine is indicated for the emergency treatment of allergic reactions (anaphylaxis) to insect bites and stings, food, medications and other allergens, as well as for idiopathic or exercise induced anaphylaxis. The EpiPen and EpiPen Jr autoinjectors are designed for the immediate self-administration of persons with a history of anaphylactic reactions. Such reactions may occur within minutes of exposure and consist of redness, apprehension, syncope, tachycardia, filiform or undetectable pulse associated with a drop in blood pressure, convulsions, vomiting, abdominal cramps and diarrhea, involuntary evacuation, wheezing, dyspnoea Due to pharyngeal spasm, pruritus, rashes, urticaria or angioedema. EpiPen and EpiPen Jr should only be used as emergency supportive therapy and do not replace or replace immediate medical or hospital care.
DOSAGETreatment with tolvaptan should be initiated at a dose of 15 mg once daily. The dose may be increased to a maximum of 60 mg once daily as tolerated to achieve the desired serum sodium level. During dose adjustment, both serum sodium and the controversial condition of the patients should be monitored. If there is no adequate increase in serum sodium levels, other treatment options should be considered either in place of tolvaptan or as an adjuvant treatment. In patients who achieve an adequate increase in serum sodium levels, they should monitor the underlying disease and serum sodium levels at regular intervals to assess whether tolvaptan treatment is necessary. In the context of hyponatremia, the duration of treatment is determined by the underlying disease and its treatment. Treatment with tolvaptan is expected to continue until the underlying disease has been adequately treated or until hyponatraemia is no longer a clinical problem.
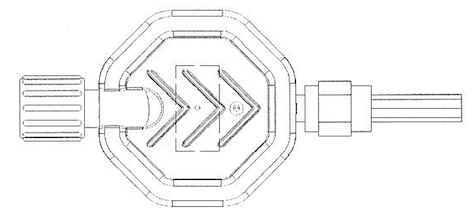
APPLICATION AREAS Intended area of application for this filter is the infusion therapy. It is to be used for the filtration on infusion solutions, protection against the contamination through bacteria, fungi, microorganisms and endotoxins up to 120h, to emit air bubbles and hold back extraneous particles. Commercial infusion sets and connection tubings with luer-lock-fittings can be connected.
APPLICATION AREASIntended area for application for this filter is the infusion therapy. It is to be used for the filtration of infusion solutions, protection against the contamination through bacteria, fungi, microorganisms and endotoxins, to emit air bubble and hold back extraneous particles. Commercial infusion sets and connection tubings with luer-lock-fittings can be connected.
Product FeaturesThe mechanism of action of HAMSYL is based on the selective killing of leukemic cells due to depletion of plasma asparagine. Some leukemic cells are unable to synthesize asparagine due to a lack of asparagine synthetase and depend on an exogenous source of asparagine for survival. The depletion of asparagine, which results from treatment with the enzyme L-asparaginase, kills the leukemic cells. Normal cells, however, are less affected by depletion because of their ability to synthesize asparagine.
THERAPEUTIC INDICATIONSDantrolene Intravenous is indicated as an appropriate supportive measure for the management of fulminant hypermetabolism of skeletal muscle characteristic of the Malignant Hypertemia crisis. This should be administrative by intravenous injection as soon as the reaction of Maltemia Hypertemia is recognized.
|
INFORMATIONThis blog is designed to provide information about acute lymphoblastic leukemia as well as treatment options and support for both patients and medical staff. All rights reserved to the group Asparaginase Pegilada. ArchivesCategories |


 RSS Feed
RSS Feed
